
*Sponsored
Market Maven Insights Initiates Coverage On (ENLV) Starting This Morning
—Monday, November 24, 2025
(ENLV) Is Backed by Multiple Potential Catalysts — Here’s What Stands Out Now
Tight Float: (ENLV) has a public float of less than 25M shares, meaning even modest interest can have an outsized impact.
Strong Clinical Results: Phase IIa results showed a 72% pain drop and strong function improvement in patients 60+, placing (ENLV) above typical benchmarks for Phase III success.
Big Market: With No Approved Disease-Modifying Therapy
Over 32M Americans suffer from osteoarthritis, and Allocetra™ targets a multi-bn-dollar space with no approved disease-modifying options, giving (ENLV) room to lead.
Analysts See Major Upside Potential: Coverage from H.C. Wainwright ($7 target) and D. Boral Capital ($13 target) suggests 650% to 1,300% potential upside.
Take a Look at (ENLV) Before the Bell Rings…
November 24, 2025
Dear Reader,
Two major announcements hit the tape from Enlivex Therapeutics (Nasdaq: ENLV) this morning — and together, they’ve pushed the company straight to the top of our radar.
First, the company released positive six-month topline results from its Phase IIa Allocetra™ trial in moderate to severe knee osteoarthritis.
The new data confirms and extends the strong three-month findings reported in August, showing durable pain reduction and meaningful functional improvement in the primary responder population aged 60 and above.
The six-month analysis again reached statistical significance, reinforcing Allocetra’s potential as a long-lasting, disease-modifying treatment in a condition with limited effective options.
On top of that, (ENLV) announced a $212M PIPE fin-ancing, priced at a premium, designed to support the company’s clinical progress while also implementing a novel treasury strategy tied to the RAIN decentralized prediction markets protocol.
Following the transaction, former Italian Prime Minister Matteo Renzi is expected to join the board — a notable addition for a company of this size.
Following both releases, (ENLV) tapped $1.75 this morning, marking an approximate 70% early move from Friday’s $0.89 range.
Momentum potential is already showing up technically as well — (ENLV) is now triggering multiple bullish signals on TradingView’s analysis dashboard, including strengthening momentum readings and key trend-line confirmations.
If you missed my earlier email, keep reading to quickly get up to speed.
There’s a moment in every breakthrough story where the data stops being “early progress” and starts hinting at something much bigger.
That moment is unfolding right now for Enlivex Therapeutics (Nasdaq: ENLV).
For years, knee osteoarthritis has been one of the most frustrating conditions in modern medicine, affecting more than 32M Americans, yet offering no approved disease-modifying therapy. Patients get temporary relief, short-term fixes, or treatments that never touch the underlying problem. The medical world has been waiting for someone to change that narrative.
(ENLV) may have taken the first real step.
With recent momentum under $1 and backed by a tight float of fewer than 25M shares, (ENLV) released Phase IIa results that didn’t just look exciting, they exceeded performance levels commonly associated with Phase III success. Pain dropped. Function improved. And most importantly, the data signaled the potential for a mechanism that goes beyond symptom management.
That’s the kind of shift that forces analysts to re-examine what they thought they knew. Some are now estimating upside scenarios ranging from 650% to 1,300% upside potential, anchored directly to the strength of those results.
A small company targeting a massive unmet medical need… suddenly showing data that could rewrite the path forward for millions.
That’s why (ENLV) isn’t just another name on a list, it’s the profile rising to the top of our radar this morning.

Rewriting the Biology of Inflammation
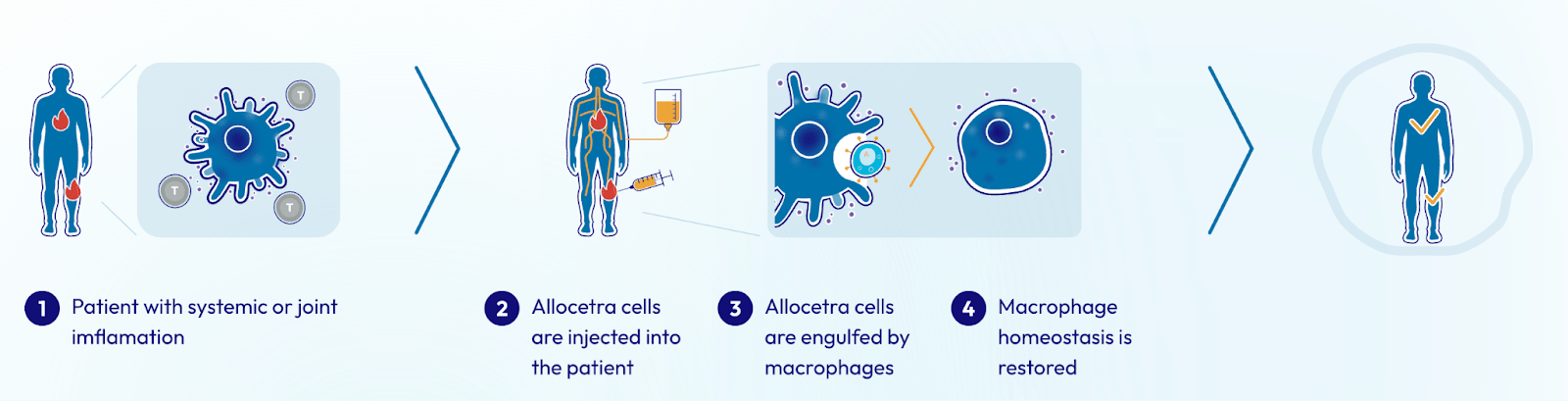
Enlivex Therapeutics (Nasdaq: ENLV) is a clinical-stage biotech company developing a new class of immune-balancing therapies designed to reset the body’s inflammatory response, not suppress it.
At the center of their platform is Allocetra™, an off-the-shelf, cell-based therapy engineered to restore immune homeostasis. Instead of targeting symptoms or blocking single pathways, Allocetra™ aims to rebalance the immune system at the source, allowing the body to correct inflammatory damage more effectively.
What Makes (Nasdaq: ENLV) Approach Stand Out
• A Platform, Not a Single Drug: Allocetra™ isn’t limited to one condition. It’s being advanced as a modular therapeutic platform with applications across multiple diseases driven by immune imbalance.
• Knee Osteoarthritis (OA): Their lead program targets knee osteoarthritis, a condition affecting more than 32 million Americans, where no approved disease-modifying therapy currently exists. (ENLV)’s Phase IIa results showed reductions in pain and meaningful improvements in function, signaling the potential to treat the underlying biology rather than manage symptoms.
• Severe Inflammation & Organ Dysfunction: (ENLV) is also developing Allocetra™ for acute conditions marked by overwhelming immune system activity, including sepsis and other life-threatening inflammatory responses where treatment options remain limited.
• Designed for Broad Clinical Utility: Because Allocetra™ works by restoring immune balance, its potential spans from chronic degenerative diseases to acute systemic inflammation, giving (ENLV) multiple shots on goal with the same foundational technology.
Inflammation sits at the center of many of the world’s most burdensome diseases. By targeting the immune system’s control center rather than individual symptoms or pathways, (ENLV)’s approach has the potential to reshape how a wide range of conditions are treated.
Recent Momentum & What’s Coming Next
(Nasdaq: ENLV) has been accelerating its clinical and strategic progress over the past few months, and several developments have started to reshape expectations around its lead program, Allocetra™.
In August 2025, the company reported positive Phase IIa data showing meaningful reductions in pain and strong functional improvement in knee osteoarthritis patients, with the 60+ subgroup demonstrating standout results.
Shortly after, (ENLV) strengthened its position with a new Israeli patent protecting the use of Allocetra™ in osteoarthritis, expanding the company’s IP footprint.
The company also laid out a clear development roadmap, with six-month follow-up data expected in November 2025 and a Phase IIb trial targeted for Q2 2026.
Most recently (ENLV) announced it would present its Phase IIa 3-month data at ACR Convergence 2025, a major rheumatology conference, increasing industry visibility at a pivotal moment.
Together, these steps show a company tightening its clinical story, expanding its IP position, and preparing for the next stage of development, all with market-moving potential catalysts on the near horizon.

Analysts Point to Significant Upside Backed by Strong Clinical Progress
Analyst coverage on Enlivex (Nasdaq: ENLV) has turned notably optimistic, driven by recent trial results and the company’s positioning in a large unmet medical market.
H.C. Wainwright reiterates a $7 target, reflecting more than 650% upside potential from recent levels near $1.03.
D. Boral Capital sets an even higher bar with a $13 target, suggesting roughly 1,300% upside if (ENLV) continues to execute on its clinical roadmap.
With fewer than 25M shares in the public float, (ENLV)’s structure adds torque to the setup, something reflected earlier this year when (ENLV) ran 130% from $0.90 to $2.10 between June 13 and August 15.
Technical Setup Brings (ENLV) Into Focus
Enlivex (Nasdaq: ENLV) is tightening up on the chart after a wide trading year, moving within a $0.81–$2.10 52-week range and currently sitting under $1.
(ENLV) saw a sharp move following the release of its positive Phase IIa knee OA data before cooling off, a common pattern in small-cap biotech as early retail activity gives way to slower institutional positioning.
With fewer than 25M shares in the public float, (ENLV) remains highly sensitive to shifts, making it a candidate for sharper moves as upcoming readouts approach.
Several milestones expected over the next six months add to the technical potential, a setup that often draws attention in low-float biotech names.
7 Reasons Why (Nasdaq: ENLV) Is Rising to the Front of Today’s Watchlist
Tight Share Structure: Fewer than 25M shares gives (ENLV) the kind of setup that can move quickly when interest turns.
Standout Mid-Stage Data: Phase IIa results showed 72% pain reduction and 109% functional improvement in patients 60+, exceeding benchmarks linked to Phase III success.
Targeting a Major Unmet Market: Knee OA affects 32M Americans, with no approved disease-modifying therapy, a market valued at $6–8B, expanding toward $14–16B.
Strong Analyst Targets: H.C. Wainwright at $7 and D. Boral Capital at $13 point to 650%–1,300% upside potential.
Clear Catalysts Ahead: Six-month data expected November 2025 and Phase IIb planned for Q2 2026.
Favorable Safety Record: Across 150+ treated patients, no serious adverse events reported, an important advantage for regulatory progress.
Positioned for Corporate Moves: The company continues exploring partnerships and non-dilutive funding, signaling potential business developments ahead of late-stage trials.
Get (ENLV) on Your Screen Before the Bell…
With a tight share structure, compelling Phase IIa data, and a massive underserved market in front of it, Enlivex (Nasdaq: ENLV) sits at a pivotal point in its development story. The combination of upcoming potential catalysts, supportive analyst coverage, and a clean safety profile creates a setup that’s difficult to ignore, especially with six-month durability data and Phase IIb progression on the horizon.
As more eyes turn toward the knee osteoarthritis space and the need for true disease-modifying therapies grows, (ENLV) is positioned to move from a quiet clinical name to a headline-driven contender.
It’s at the top of our early watchlist as the session approaches.
Keep it on your screen—another update is coming.
Sincerely,
Tate Remington
Chief Editor, Market Maven Insights
MarketMavenInsights.com (“MarketMavenInsights” or “MMI”) is owned by Source Coastal Media LLC, a multi member limited liability company. Data is provided from third-party sources and MMI is not responsible for its accuracy. Make sure to always do your own research and due diligence on any day and swing profile MMI brings to your attention. Any emojis used do not have a specific defined meaning, and may be used inconsistently. We do not provide personalized in.vest.ment advice, are not in.vest.ment advisors, and any profiles we mention are not suitable for all in.vest.ors.
Pursuant to an agreement between Source Coastal Media LLC and TD Media LLC, Source Coastal Media LLC has been hired for a period beginning on 11/24/2025 and ending on 11/24/2025 to publicly disseminate information about (ENLV:US) via digital communications. Under this agreement, Source Coastal Media LLC has been paid seven thousand five hundred USD (“Funds”). These Funds were part of the funds that TD Media LLC received from a third party who did not receive the Funds directly or indirectly from the Issuer and does not own stock in the Issuer but the reader should assume that the clients of the third party own shares in the Issuer, which they will liquidate at or near the time you receive this communication and has the potential to hurt share prices.
Neither Source Coastal Media LLC, TD Media LLC and their member own shares of (ENLV:US).
Please see important disclosure information here: https://marketmaveninsights.com/disclosure/enlv-mKNGF/#details